He came up with theories about how atoms combine to make compounds and also came up with the first set of chemical symbols for the known elements.ĭalton’s outlining of atomic theory was a start, but it still didn’t really tell us much about the nature of atoms themselves. However, since the neutron wouldn’t be discovered until 1932, we can probably forgive Dalton for this oversight. The latter point is one that pretty much still holds true, with the notable exception being isotopes of different elements, which differ in their number of neutrons. 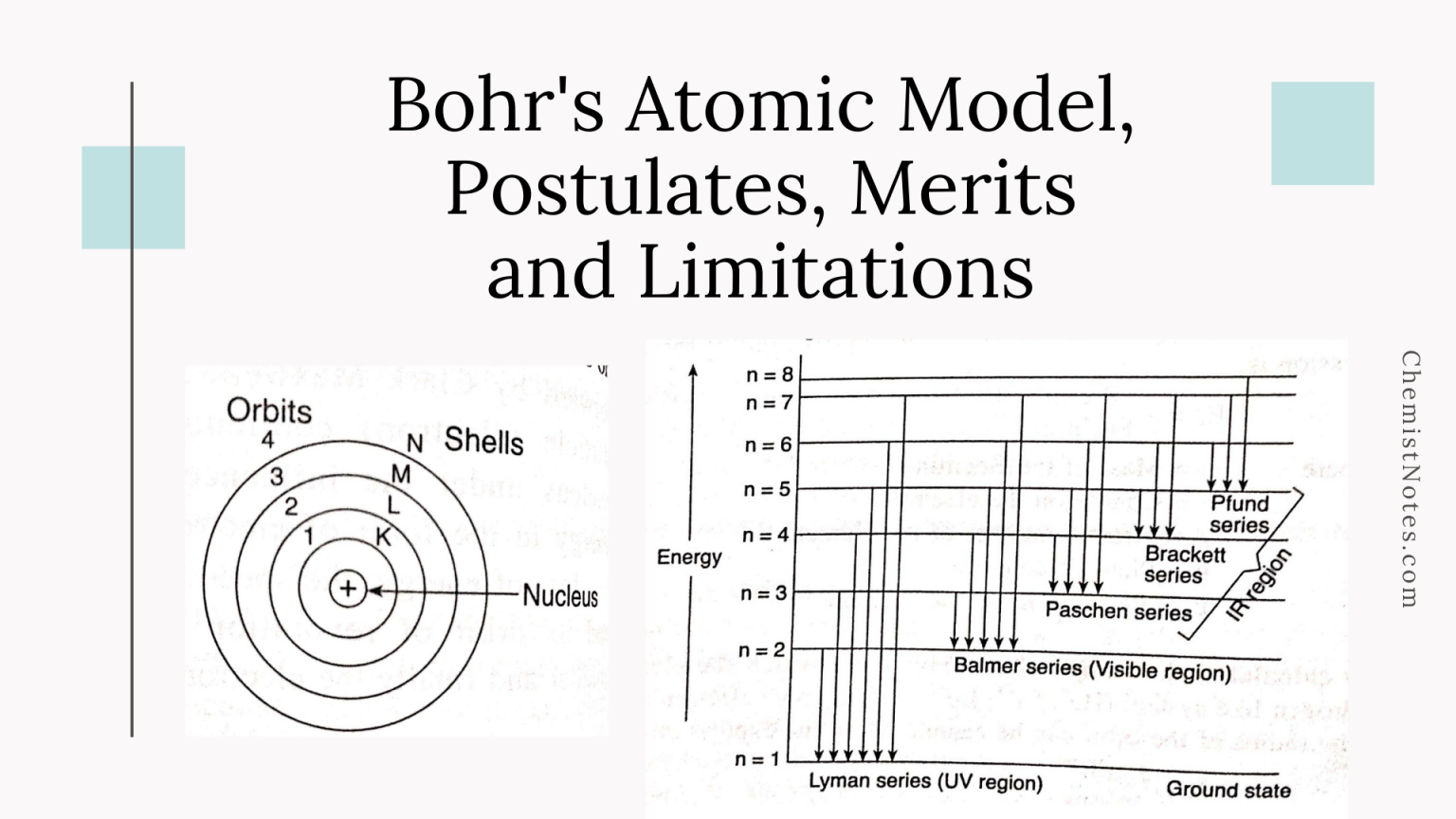
He drew on the ideas of the Ancient Greeks in describing atoms as small, hard spheres that are indivisible, and that atoms of a given element are identical to each other. It wasn’t until 1803 that the English chemist John Dalton started to develop a more scientific definition of the atom. It was a long wait, however, before these foundations were built upon. Though we now know that this is not the case, their ideas laid the foundations for future atomic models. Water atoms were smooth and slippery, explaining why water was a liquid at room temperature and could be poured. They envisaged iron atoms as having hooks which locked them together, explaining why iron was a solid at room temperature. These scholars imagined atoms as varying in shape depending on the type of atom. Though their ideas about atoms were rudimentary compared to our concepts today, they outlined the idea that everything is made of atoms, invisible and indivisible spheres of matter of infinite type and number. The Ancient Greek theory has been credited to several different scholars but is most often attributed to Democritus (460–370 BC) and his mentor Leucippus. The word ‘atom’ actually comes from Ancient Greek and roughly translates as ‘indivisible’. In fact, we have to go all the way back to Ancient Greece to find its genesis. Though our graphic starts in the 1800s, the idea of atoms was around long before. This graphic takes a look at the key models proposed for the atom, and how they changed over time. Despite this, our ideas about what an atom is are surprisingly recent: as little as one hundred years ago, scientists were still debating what exactly an atom looked like. This is something we now take as a given and one of the things you learn right back at the beginning of high school or secondary school chemistry classes. Therefore, these orbits are also known as energy levels or energy shells.All matter is made up of atoms. Each orbit is associated with a fixed amount of energy. We should note that the nucleus contains all the protons and neutrons of the atom. 
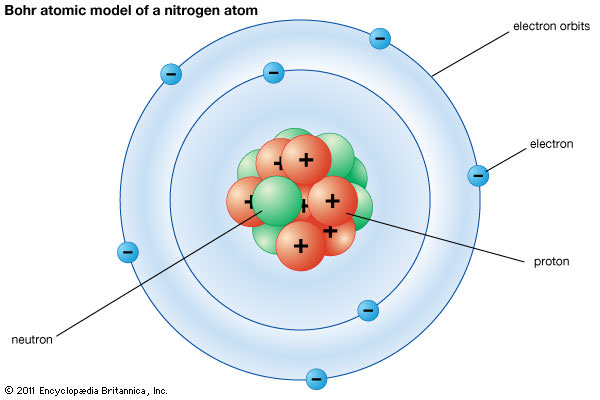
And the whole mass of the atom is concentrated at the nucleus. Note: Bohr Theory also states that the atom consists of a small positively charged nucleus at its centre. So, from the above discussion we know about the Bohr Model of atoms. We should note that electrons can only gain or lose energy by jumping from one allowed orbit to another, absorbing or emitting electromagnetic radiation with a frequency (ν) determined by the energy difference of the levels according to the Planck relation. In these orbits, an electron’s acceleration does not result in radiation and energy loss as required by classical electromagnetic theory. These orbits are associated with definite energies and are also called energy shells or energy levels. They orbit at a certain discrete set of distances from the nucleus. In the second point it states that electrons can only orbit stable orbits without radiating. We should note that the Bohr Model states that electrons in atoms orbit around the nucleus. Now, we should know about some properties of electrons under the Bohr Model. The Bohr Theory solved this problem and correctly explained the experimentally obtained Rydberg formula for emission lines. These planetary models ultimately predicted all atoms to be unstable due to the orbital decay. In terms of electron emission, we can say that a continuous amount of frequencies being emitted and the electron moved closer to the nucleus, it would move faster and would emit a different frequency than those experimentally observed. We should know that early planetary models of the atom suffered from a flaw: in this model electrons spinning in orbit around a nucleus. Now, we should know about the need for the Bohr model of atoms. We should know that Bohr model of atom was the further development is the earlier cubic model (1902), the plum-pudding model (1904), the Saturnian model (1904), and the Rutherford model (1911). Hint: To correctly describe Bohr’s model of atom, we should know that Bohr’s model of atom states that electrons in atoms move in circular orbits around a central nucleus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
